The U.S. Styrene Information Study Committee recently released the results of a study. The study was conducted on nearly 16,000 workers who were exposed to styrene at work. The results showed no conclusive evidence that styrene increases the risk of cancer.
The study, “Cancer Mortality among Styrene Workers in the U.S. Reinforced Plastics and Composites Industry,” was published in the March 2013 issue of the scientific journal Epidemiology. The research committee said they were a funder of the study, but had no involvement in its content.
The authors of this study investigated mortality due to cumulative exposure, continuous exposure, peak exposure, average exposure, and number of exposures after first exposure to styrene, based on 60 years of epidemiological information.
This is the second study to declare no relationship between styrene exposure and cancer. Cambridge, Massachusetts-based environmental and risk science consulting firm Gradient, Inc. analyzed the extant styrene research literature. Its results also showed no relationship between styrene exposure and increased mortality due to any type of cancer.
From June 2011 to the present, the Styrene Information Study Committee and its member companies have been seeking strong evidence against the inclusion of styrene on the list of “reasonably anticipated carcinogens” in the National Toxicology Program’s (NTP) Phase 12 report.
On November 11, 2011, the U.S. Department of Health released a new report showing that the government has officially added styrene and seven other chemicals to the list of substances that may cause cancer in humans. Styrene is widely used in plastic packaging, disposable paper cups, food containers and construction materials.
Due to strong opposition from manufacturers, the U.S. government has delayed the inclusion of these hazardous substances on the “carcinogenic list” for several years.
Formaldehyde is also on the list of this inclusion. The report further highlights the dangers of formaldehyde. The report says formaldehyde is recognized as a carcinogen capable of causing certain types of leukemia. It is present in plywood, cardboard, and even some hair care products.
The report states that the warnings for the listed carcinogens came from studies of workers in industrial settings. Most workers are exposed to these substances in the course of their work.
Philip Landrigan, dean of the Mount Sinai School of Medicine School of Global Health in New York, recommends that people, especially pregnant women and children, should avoid polystyrene containers, as well as other products that use styrene.
The warning was based on a report from the National Academy of Toxicology and some of the National Institutes of Health on carcinogens. This is the 12th time that the report has been issued, and the last time it was issued was in 2005.
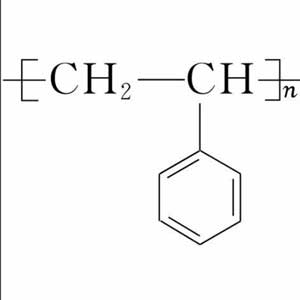
Styrene, an organic compound with the chemical formula C8H8. The electrons of vinyl are conjugated to the benzene ring. It is insoluble in water and soluble in most organic solvents such as ethanol and ether. Styrene is an important monomer for synthetic resins, ion exchange resins and synthetic rubber, etc. On October 27, 2017, the World Health Organization’s International Agency for Research on Cancer published a preliminary list of carcinogens with reference to styrene in the list of carcinogens in category 2B.
Styrene is a colorless, oily liquid with an aromatic odor. Its freezing point is minus 30.6°C. Boiling point is 145°C. Relative density is 0.9051. styrene’s flash point (open cup) is 31.11℃. The auto-ignition point is 490°C. Refractive index is 1.5467. Viscosity (20°C) 0.763 mPa -s.
Styrene in the heat or exposure to sunlight, or in the presence of peroxide is easy to polymerization, polymerization of heat release, and can cause an explosion. Explosion limit 1. 1% ~ 6. 1% (by volume).
Styrene is mainly used as an important monomer for synthetic resins, ion exchange resins and synthetic rubber, etc. It can also be used in pharmaceutical, dye, pesticide and mineral processing industries.
Styrene is a serious hazard to the environment. Special attention should be paid to the pollution of surface water, soil, atmosphere and drinking water. Styrene should be given special attention to aquatic organisms.
Because of its volatility, it is easily photolyzed in the atmosphere, and can also be biodegraded and chemically degraded, i.e., it can be destroyed by specific bacteriological clusters, and can also be oxidized by oxygen in the air to anisole, formaldehyde and small amounts of phenethyl alcohol.
The U.S. Styrene Information Study Committee said the U.S. National Toxicology Research Program relied on biased and erroneous available data. And the European Union has rejected styrene as a carcinogen after a review of the same information.
Although styrene has no relationship with cancer, higher concentrations of styrene can still put exposed individuals at risk.
A report on the real-life treatment of patients with styrene poisoning is as follows.
1. Quick response, accurate diagnosis, and correct first aid measures are guarantee of successful treatment.
After the hospital received the call, a monitoring ambulance was urgently activated. Medical personnel arrived at the scene within 10 minutes. On the way to the hospital, communicate information with the emergency department staff in the hospital. Pre-hospital emergency care is provided at the same time.
According to the clinical manifestations, the patients are classified into three categories: mild, moderate, and severe. The patients were placed in the resuscitation room and the emergency room respectively. Keep the respiratory tract open, administer oxygen, open intravenous access, and implement the first aid plan one by one.
2. Prevent secondary contamination in the first aid process and pay attention to the safety and protection of medical personnel.
2.1 Remove the source of contamination
When the patient enters the emergency area, the air is filled with the smell of styrene volatilized. Nurses put on rubber gloves, first remove the contaminated clothes from the patients, put them into a closed plastic bag, and move them out of the emergency area for separate treatment.
2.2 Ventilation
Ensure air circulation in the emergency room. Due to the increase of styrene content in the air of the emergency room caused by the exhalation of gas from the mouth of the poisoned patient, increase the air convection in the room to effectively prevent the medical staff from inhaling the styrene dispersed in the air.
2.3 Timely change of treatment personnel
When the patient arrives at the emergency area, quickly start the hospital public accident treatment plan. The emergency echelon personnel arrived on standby. The hospital’s chief medical officer will unify the deployment. The medical personnel in the emergency room are changed every 30 minutes and all medical personnel entering the emergency area are required to wear double masks.
2.4 Strengthen the safety protection work in the hospital room
After the patient is sent to the ward after the emergency period, ventilation is maintained in the ward. Medical personnels wear double masks when entering to avoid secondary contamination of medical personnel. All medical personnel who participated in the rescue should change their uniforms, masks, and work caps in time after work, and wash their hands thoroughly with running water.
3. Summary.
3.1 During the rescue process, because of effective protective measures, timely removal of contaminated clothing, and a good job of ventilation in the emergency area, effectively prevented the secondary contamination of medical personnel by styrene.
3.2 Strengthening health knowledge publicity and education, strict compliance with operating procedures, improving the awareness of prevention, do a good job of occupational hazard protection.