Industrial microbial contamination is the primary cause of system corrosion, efficiency decline, product spoilage, and shutdowns for cleaning. Traditional oxidative biocides suffer from significant pH sensitivity, prone to byproduct formation, equipment corrosion, and poor compatibility issues; while conventional non-oxidative biocides exhibit drawbacks such as slow onset, high residues, and poor biofilm penetration. DBNPA-based biocides have emerged as the mainstream choice in the global industrial biocide market due to their rapid killing action, high efficacy at low doses, broad-spectrum coverage, rapid degradation, and environmental friendliness.
This paper systematically analyzes the core performance boundaries of DBNPA biocides from a molecular structural perspective. It clarifies the mechanism of action, pH dependency patterns, degradation pathways, quantitative analysis, formulations, and applications, providing a comprehensive technical framework for engineering and R&D.
1. Mechanism of Action of DBNPA Biocide
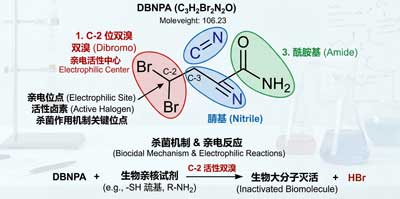
The mechanism of action for DBNPA biocide is non-oxidative and electrophilic in nature, differing from broad-spectrum oxidative destruction agents like chlorine gas and sodium hypochlorite. It offers advantages of more precise targeting, faster onset of action, and better compatibility.
1.1 Penetration and Entry
With its small molecular size and moderate lipophilicity, DBNPA rapidly penetrates the cell membranes of bacteria, fungi, and algae to exert its intracellular effects. It also demonstrates excellent penetration and stripping capabilities against biofilms, effectively addressing slime issues that conventional fungicides struggle to eliminate.
1.2 Core Targets
Targets: sulfur-containing amino acids (cysteine, methionine). The C-2 position of DBNPA exhibits strong electrophilicity due to its dibrominated substitution, preferentially forming irreversible covalent bonds with sulfhydryl (-SH) groups and thioether bonds on enzyme proteins. This inactivates key enzymes involved in the respiratory chain, ATP synthesis, and substance transport, causing cellular energy metabolism to collapse.
Disruption of the redox system blocks normal cellular oxidation-reduction cycles, interrupting electron transport chains and causing microbial death within minutes.
Bromination Modification and Enzyme Inactivation Selectively brominates key microbial metabolic enzymes and coenzymes, inhibiting nucleic acid and protein synthesis to achieve dual bactericidal and algal-suppressing effects.
1.3 Bactericidal Kinetic Characteristics
- High efficacy at low concentrations (0.5–20 mg/L).
- Bactericidal rate >80% after 30 min exposure; >90% after 60 min; >95% after 90 min.
- Effective against Gram-positive/negative bacteria, yeast, fungi, algae, and sulfate-reducing bacteria (SRB).
2. pH Value and Stability Patterns of DBNPA
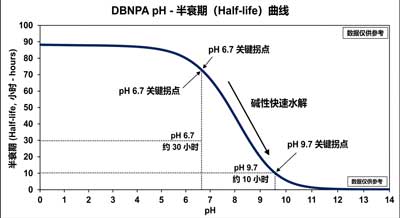
The pH value of DBNPA directly determines the chemical’s effective lifespan, dosing strategy, and system compatibility, making it a core parameter in engineering design.
2.1 Relationship Between pH and Stability
| pH Value | Stability | Half-Life (25°C) | Applications |
|---|---|---|---|
| 4.0–6.0 | Excellent | >100 h | Acidic water treatment, electroplating solutions, acidic cutting fluids |
| 6.0–7.5 | Good | 15–37 h | Circulating water, RO feedwater, neutral pulp |
| 7.5–8.5 | Fair | 1–5 h | Weakly alkaline water (requires rapid shock dosing) |
| >9.0 | Poor | <0.5 h | Continuous dosing not recommended |
2.2 Engineering Suggestions
- Optimal dosing range: pH 4.0–8.0.
- For high pH systems (pH > 8.5), shock dosing is recommended to leverage the “disinfection-first, degradation-second” mechanism for efficient control.
- Avoid coexistence with strong alkalis or strong reducing agents (e.g., H2S) to prevent rapid deactivation due to bromine loss.
3. DBNPA Degradation Process and Pathways
The DBNPA degradation process is the core enabler of its “high efficiency + environmental friendliness”: rapid breakdown into non-toxic small molecules after disinfection, leaving no persistent residues and meeting discharge requirements.
3.1 Primary Decomposition Triggers
- High pH (dominant factor)
- Temperature increase
- UV/visible light
- Reducing substances (H₂S, sulfites, etc.)
- Microbial enzymatic decomposition
3.2 Typical Degradation Pathways
DBNPA degradation in aqueous phase follows the sequence:
- de-bromination → hydrolysis → mineralization.
- Dibromination yields a monobrominated intermediate: 2-bromo-3-cyanopropionamide.
- Hydrolysis produces dibromoacetonitrile, dibromoacetamide, cyanoacetamide, bromoacetic acid, etc.
- Ultimately mineralized into: Br–, NH4+, CO2, H2O intermediate and final products are low-toxicity/non-toxic.
3.3 Engineering Value of Degradation
- Rapid inactivation post-disinfection, preventing downstream microbial inhibition.
- Discharge-friendly, suitable for zero-discharge/reuse systems.
- Safe for use with sensitive units like RO membranes and ion exchange resins.
4. DBNPA Analysis (Quantitative and Quality Control Methods)
DBNPA analysis is an essential tool for formulation quality control, on-site dosing, and residue monitoring. The following are reliable industrial standard methods.
4.1 High-Performance Liquid Chromatography (HPLC, Preferred)
- Column: C18
- Mobile Phase: Methanol / Water
- Detection Wavelength: 215–220 nm
- Advantages: High accuracy, strong interference resistance, simultaneous detection of intermediates and degradation products.
- Applications: Active ingredient content, formulation content, water sample residues.
4.2 Ultraviolet Spectrophotometry
- Characteristic Absorption: ~218 nm
- Applications: Rapid screening, on-site rapid testing.
- Limitations: Significant interference from complex matrices.
4.3 Chemical Titration Method (Indirect Bromine Value/Bromine Index Approach)
Utilizes DBNPA’s bromine release characteristic to indirectly estimate active content via titration. Suitable for rapid in-plant monitoring.
4.4 Quality Control Specifications (Typical Industrial Grade 99% Specification)
- Appearance: White crystalline powder
- Purity: ≥99.0%
- Melting Point: 122–126 °C
- Loss on Drying: ≤0.5%
- pH of 1% Aqueous Solution: 5.0–7.0
- Individual Impurities: ≤0.5%
5. Based DBNPA Biocides: Formulation and Dosage Form Design
Pure DBNPA is rarely used in industrial applications. Formulated DBNPA biocides are predominantly marketed to enhance solubility, stability, compatibility, and biofilm penetration.
5.1 Common Dosage Forms
Water-Soluble Liquid Formulations (10%–50%)
- Solvent: Water + Co-solvent (ethylene glycol, propylene glycol, PEG, etc.)
- Advantages: Easy dosing, dust-free, compatible with automated dosing systems.
Powder Formulations
- Blended with dispersants, stabilizers, and defoamers
- Suitable for paper manufacturing and powder coatings applications
5.2 Synergistic Formulation Strategies (Enhanced Efficacy Without Antagonism)
- DBNPA + Isothiazolinone: Broad-spectrum, long-lasting
- DBNPA + Tetraalkylphosphonium salt: Enhances stripping and algae inhibition
- DBNPA + Glutaraldehyde: Treats severe slime and biofilms
- DBNPA + Dispersant: Prevents re-adhesion
5.3 Formulation Contraindications
- Prohibited from long-term coexistence with strong oxidizing/reducing systems.
- High-alkaline systems require immediate on-site application.
- Avoid excessive mixing with amines (accelerates decomposition).
6. Industrial Application Scenarios and Dosage Guidelines
DBNPA-based biocides cover nearly all aqueous industrial systems:
1. Industrial Circulating Cooling Water
- Dosage: 5–20 mg/L (shock/continuous)
- Functions: Bactericidal, algal inhibition, slime stripping, enhanced heat transfer efficiency, corrosion reduction.
2. Reverse Osmosis (RO)/Nanofiltration Membrane Systems
- Non-oxidizing, low residue, non-damaging to polyamide membranes.
- Dosage: 0.5–5 mg/L, online bacterial inhibition.
3. Paper Mill White Water / Pulp
- Controls sludge, resin blockage, staining, and paper breaks
- Foam-free, does not affect strength or brightness
4. Oilfield Injection Water / Fracturing Fluids
- Highly effective against SRB, controls hydrogen sulfide and corrosion.
- Rapid degradation, harmless to formation microorganisms.
5. Metalworking Fluids / Cutting Fluids / Emulsions
- Extends service life, prevents odor and spoilage.
- Compatible with pH 5–8 systems.
6. Paints, Wood, Adhesives, Leather
- Anti-corrosion and anti-mold, low toxicity and environmentally friendly
Conclusion
DBNPA-based biocides are broad-spectrum, fast-acting, low-residue, environmentally friendly non-oxidizing industrial biocides suitable for the vast majority of aqueous industrial systems.
The mechanism of action of DBNPA biocides centers on electrophilic attack on sulfhydryl enzymes, blocking redox reactions, and rapid lethality, demonstrating high efficacy against both free-floating microorganisms and biofilms.
pH is a critical control parameter for DBNPA: optimal performance occurs at pH 4–8, while alkaline conditions cause rapid degradation. This property enables dual objectives of “biocidal efficacy and self-degradation.”
The DBNPA degradation process is clearly controllable, ultimately mineralizing into non-toxic products that meet environmental and reuse requirements.
HPLC serves as the standard analytical method for DBNPA, enabling precise quantification of the active ingredient, formulations, and water samples.
Formulated DBNPA biocides offer irreplaceable comprehensive advantages in applications such as circulating water, RO systems, papermaking, oilfields, and metalworking fluids.
References
- ChemicalBook. 2,2-Dibromo-3-cyanoacrylamide: Physical and Chemical Properties and Biocidal Mechanism. 2024.
- Water Treatment Chemicals Network. DBNPA Product Details and Industrial Applications. 2025.
- Review of Industrial Biocides Technology. DBNPA Degradation Pathways and pH Stability. 2026.
- Non-Oxidizing Biocide DBNPA: Characteristics and Application Summary. 2026.
US EPA & EU BPR. DBNPA Biocide Evaluation Report. - Chemical Industry Standards. Industrial 2,2-Dibromo-3-cyanopropionamide (HG/T Relevant Specifications).